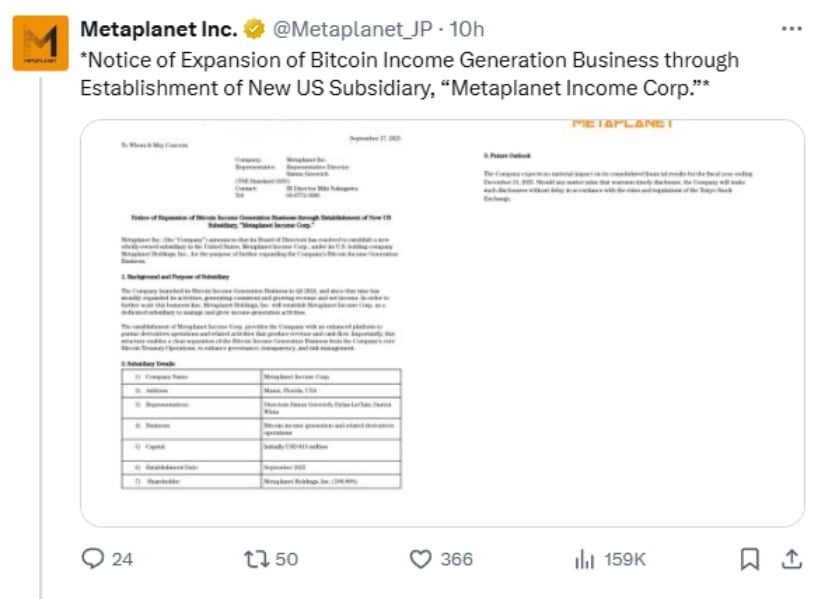
Antengene Expands XPOVIO® Indications in Malaysia with Approval in Diffuse Large B-cell Lymphoma
SHANGHAI and HONG KONG, Dec. 16, 2025 /PRNewswire/ — Antengene Corporation Limited (“Antengene”, SEHK: 6996.HK), a leading innovative, commercial-stage global biotech company dedicated to discovering, developing and commercializing first-in-class and/or best-in-class medicines for autoimmune disease, solid tumors and hematological malignancies indications, today announced that the Malaysian National Pharmaceutical Regulatory Agency has approved a supplemental New Drug Application (sNDA) for XPOVIO® (selinexor) for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) after at least 2 lines of systemic therapy, who are ineligible for autologous stem cell transplant.
With this recent approval, XPOVIO® has further expanded its portfolio of approved indications in Malaysia, bringing the total to three indications across multiple myeloma (MM) and DLBCL, two major therapeutic areas in hematology. Prior to this, XPOVIO® was approved in Malaysia for two indications, including in combination with bortezomib and dexamethasone for the treatment of adult patients with MM who have received at least one prior therapy; and in combination with dexamethasone for the treatment of adult patients with MM who have received at least four prior therapies and whose disease is refractory to at least two proteasome inhibitors, two immunomodulatory agents and an anti-CD38 monoclonal antibody, and who have demonstrated disease progression on the last therapy. The latest indication expansion into DLBCL will allow XPOVIO® to benefit a broader population of patients, offering new hope and a meaningful treatment option for patients and families who have been urgently seeking effective therapies.
With a novel mechanism of action, XPOVIO® is the world’s first approved orally-available, selective XPO1 inhibitor, which has already been approved in ten countries and regions in APAC, and has been included in the national insurance schemes in five of these markets (the mainland of China, Taiwan market, Australia, Singapore and South Korea). Moving forward, XPOVIO® is expected to receive public insurance coverage in more APAC markets.
About Antengene
Antengene Corporation Limited (“Antengene”, SEHK: 6996.HK) is a global, R&D-driven, commercial-stage biotech company focused on developing first-in-class/best-in-class therapeutics for diseases with significant unmet medical needs. Its pipeline spans from preclinical to commercial stages and includes several in-house discovered programs, including ATG-022 (CLDN18.2 ADC), ATG-037 (oral CD73 inhibitor), ATG-101 (PD-L1 × 4-1BB bispecific antibody), ATG-031 (CD24-targeting macrophage activator), and ATG-042 (oral PRMT5-MTA inhibitor).
Antengene has also developed AnTenGager , a proprietary T cell engager 2.0 platform featuring “2+1” bivalent binding for low-expressing targets, steric hindrance masking, and proprietary CD3 sequences with fast on/off kinetics to minimize cytokine release syndrome (CRS) and enhance efficacy. These characteristics support the platform’s broad applicability across autoimmune disease, solid tumors and hematological malignancies indications.
, a proprietary T cell engager 2.0 platform featuring “2+1” bivalent binding for low-expressing targets, steric hindrance masking, and proprietary CD3 sequences with fast on/off kinetics to minimize cytokine release syndrome (CRS) and enhance efficacy. These characteristics support the platform’s broad applicability across autoimmune disease, solid tumors and hematological malignancies indications.
To date, Antengene has obtained 32 investigational new drug (IND) approvals in the U.S. and Asia, and submitted new drug applications (NDAs) in 11 Asia Pacific markets. Its lead commercial asset, XPOVIO® (selinexor), is approved in Mainland of China, Taiwan China, Hong Kong China, Macau China, South Korea, Singapore, Malaysia, Thailand, Indonesia and Australia, and has been included in the national insurance schemes in five of these markets (Mainland of China, Taiwan China, Australia, South Korea and Singapore).
Forward-looking statements
The forward-looking statements made in this article relate only to the events or information as of the date on which the statements are made in this article. Except as required by law, we undertake no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, after the date on which the statements are made or to reflect the occurrence of unanticipated events. You should read this article completely and with the understanding that our actual future results or performance may be materially different from what we expect. In this article, statements of, or references to, our intentions or those of any of our Directors or our Company are made as of the date of this article. Any of these intentions may alter in light of future development. For a further discussion of these and other factors that could cause future results to differ materially from any forward-looking statement, please see the other risks and uncertainties described in the Company’s Annual Report for the year ended December 31, 2024, and the documents subsequently submitted to the Hong Kong Stock Exchange.
For more information, please contact:
Investor Contacts:
Donald Lung
E-mail: donald.lung@antengene.com
BD Contacts:
Ariel Guo
E-mail: ariel.guo@antengene.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/antengene-expands-xpovio-indications-in-malaysia-with-approval-in-diffuse-large-b-cell-lymphoma-302643379.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/antengene-expands-xpovio-indications-in-malaysia-with-approval-in-diffuse-large-b-cell-lymphoma-302643379.html
SOURCE Antengene Corporation Limited
You May Also Like

The Role of Reference Points in Achieving Equilibrium Efficiency in Fair and Socially Just Economies

Cryptos Signal Divergence Ahead of Fed Rate Decision